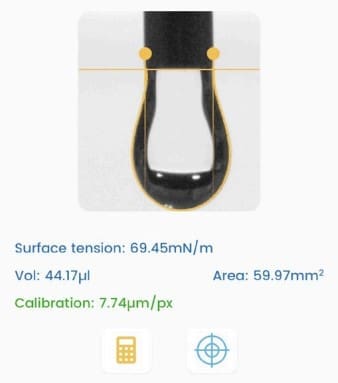
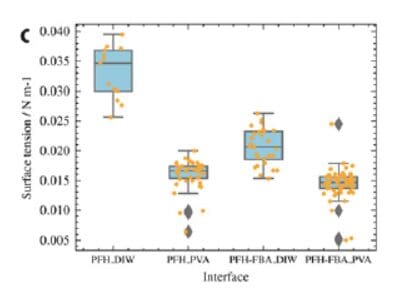
The Droplet Lab tensiometer was used in pendant-drop mode to quantify interfacial tension between perfluorohexane (PFH) and aqueous phases with/without additives. This measurement verified how the new fluorophilic boronic acid (FBA) copolymer and poly(vinyl) alcohol (PVA) each influence interfacial tension and supported the key conclusion that lasting emulsion stability requires the combined FBA–PVA interfacial assembly, not just tension reduction alone (methods: pendant-drop tensiometry; results summarized and visualized in Fig. 2b–c).